Renu Suresh
Expert
Published on: Apr 20, 2026
Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19
The
Directorate General of Foreign Trade (DGFT) has specified the procedure and criteria for exporting PPE Medical coveralls for COVID-19 vide Trade Notice No. 17/2020-21. The DGFT had earlier banned the export of PPE medical coveralls. With this notification, the DGFT has amended the Export Policy of Personal Protective Equipment (PPE), and PPE Medical Coveralls for COVID-19 is categorized as Restricted Export Item (Non-SCOMET). Hence the exporter needs to obtain export authorization from the Government for shipments of PPE Medical Coveralls for COVID-19. The exporter can apply for export authorization/license online through the official website of DGFT under the Non-SCOMET Restricted items sections.The Gist of Trade Notification
The Trade Notice on 29th June 2020 has outlined the detailed Procedure and Criteria for submitting and approving applications for the export of PPE for COVID-19.
- The DGFT has announced that the PPE Medical Coveralls for COVID-19 are in the restricted category, making it mandatory for exporters to get a license from DGFT to export the product.
- As per the trade notice, the DGFT also confirmed that the export of only 50 lakh units of PPE medical coveralls for COVID -19 would be allowed every month.
- The exporter can apply for export authorization as Non-SCOMET Restricted items and exports are not required to forward any hard copy of the application via email or post to DGFT.
Note on Restricted Export Item (Non-SCOMET)
All goods, and export permitted only with an Authorization/License following the procedure prescribed in India’s Foreign Trade Policies are known as Restricted Export Items (Non-Special Chemicals, Organisms, Materials, Equipment, and Technology (SCOMET)). For the import of restricted items, a license/authorization will have to be obtained from the DGFT.
Export of PPE Medical Coveralls
DGFT, with a Notification on 31st January 2020 had prohibited the Export of PPE, including Clothing and Masks Further with Notifications on 19th March, 16th May, and 22nd June 2020, export on the following were prohibited under the Export Policy 2018:
- Surgical Masks/Disposable Masks
- All Masks except non-surgical/non-medical masks of all types (cotton, silk, wool, knitted)
- PPE exported either as part of kits or as individual items :
- Medical Coverall of all categories
- Medical Goggles
- All Masks except non-surgical/non-medical masks of all types(cotton, silk, wool, polyester, nylon, rayon, viscose-knitted, woven or blended)
- Nitrile/NBR Gloves
- Face Shield
Restricted Export of PPE Medical Coveralls
Considering the demand for PPE during the COVID-19 pandemic, the Government revised the export policy and allowed the export of the following products with a license.
|
Sl.No |
ITC HS Code |
Description |
|
1 |
392690 | Surgical Masks Disposable Masks All Masks except non-surgical/non-medical masks of all types (cotton, silk, wool, knitted |
|
2 |
621790 | |
| 3 |
630790 |
|
| 4 |
901890 |
Note: All other items remain prohibited for export, and the monthly quota will not apply to such items.
Monthly Export Quota for PPE Medical Coveralls Export
As mentioned above, a monthly export quota of 50 Lakh PPE Medical Coveralls for COVID-19 units has been fixed, and the application filed from the 1
st to the 3rd day of each month will be considered for that month's quota. All applications for the export of PPE Medical Coveralls will be examined as per the Handbook Procedure, and all approval of the application will be done by the 10th of every month. Note: Only one application per Import Export Code will be considered during a MonthThe validity of Export Authorization/License
The validity of the Export Authorization/License for the export of PPE Medical Coveralls for COVID-19 is three months from the date of issue.
Eligibility Criteria to Obtain Export License
The firm that applies for the export license should be the manufacturer of Personal Protective Equipment (PPE) Coveralls.
Documents Required for Export License
The exporter must furnish the following documents to obtain export authorization for the shipment of PPE medical coveralls.
- Certificate of Registration as Manufacture of PPE from the concerned Department
- Copy of Testing or Accreditation of PPEz medical coveralls issued to the firm from the laboratories recognized by the Ministry of Textiles.
- Copy of importing country’s PPE medical coveralls Standard Certificate or Copy of the Bureau of Indian Standard (BIS) Certificate if the importing country does not insist on a Standard Certificate.
- Chartered Engineer’s Certificate certifying that the fabrics used in the PPE medical coverall were manufactured in India
- Copy of the Import Export Code of the Firm
- Copy of Purchase order or Invoice
Note: All documents must be duly self–attested by the authorized person of the firm.
Procedure to Obtain Export Authorization of Non-SCOMET Restricted items
The procedure to get the license for the Export of PPE Medical Coveralls for COVID-19 is given below: The exporter needs to access the official website of the
Directorate General of Foreign Trade (DGFT) to apply. Click on the service option from the home page and select the online ECOM Application. Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - DGFT Home Page
The link will redirect to the ECOM application page; click on the Restricted Export Item (Non-SCOMET) to apply for the license.
Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - DGFT Home Page
The link will redirect to the ECOM application page; click on the Restricted Export Item (Non-SCOMET) to apply for the license.
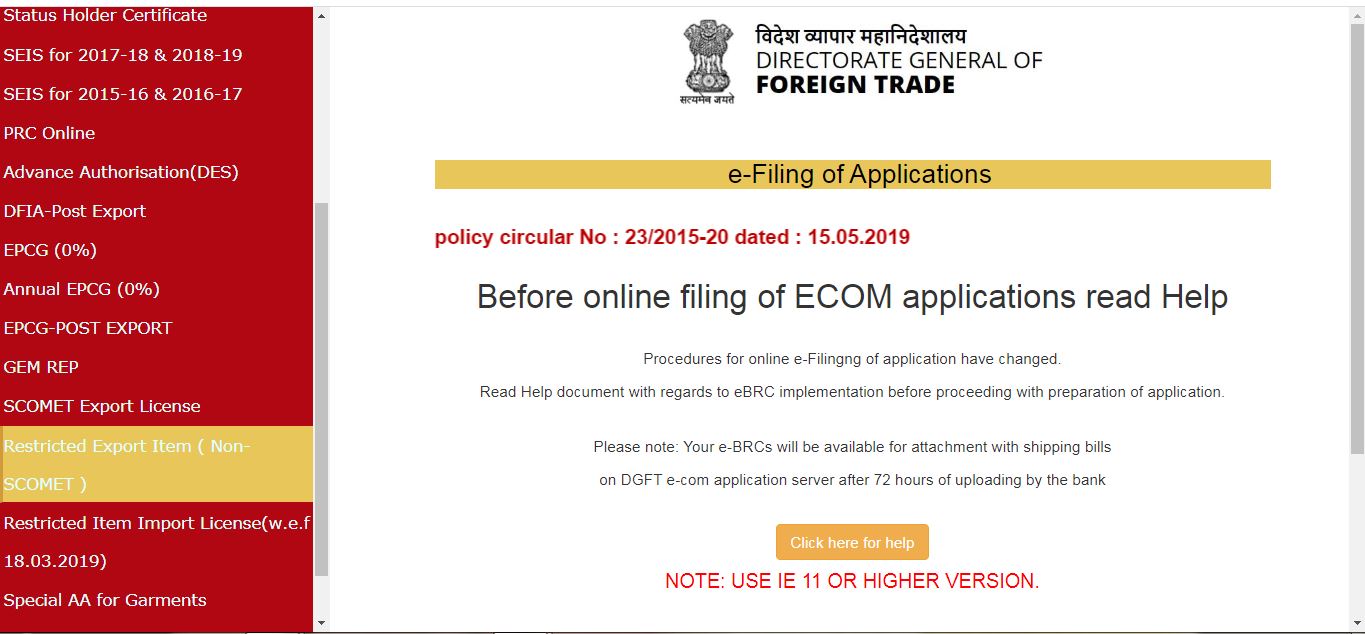 Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - ECOM
After successful login using the ECOM application module through Digital Certificate on DGFT website, 20 digits ECOM Reference number will be displayed.
Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - ECOM
After successful login using the ECOM application module through Digital Certificate on DGFT website, 20 digits ECOM Reference number will be displayed.
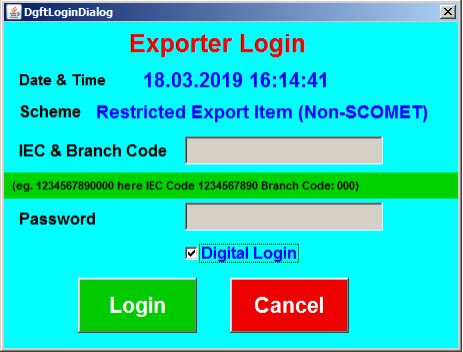 Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - DGFT Login
By clicking on the OK button, the exporters’ profile and status of an application filed in the last year will be displayed.
Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - DGFT Login
By clicking on the OK button, the exporters’ profile and status of an application filed in the last year will be displayed.
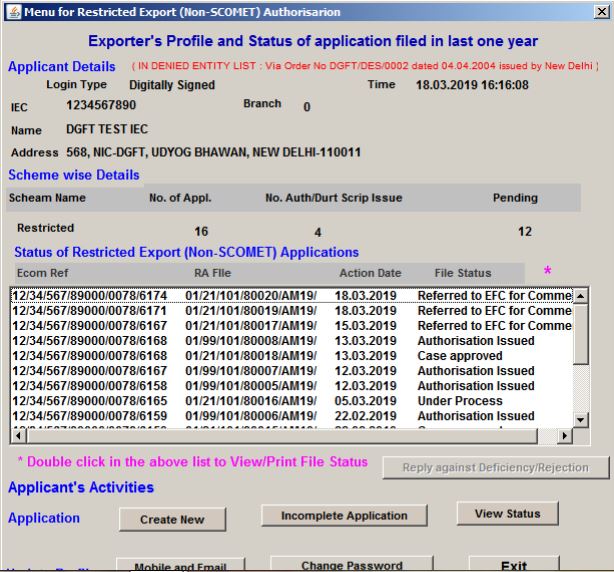 Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Exporter Profile
Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Exporter Profile
Shipment Details
From the File menu, select the Create option. A message box will appear; after confirming the details click on the ok button. The exporter can now provide details of shipment in the license application:
- Telephone Number
- Fax details
- Port of Loading/shipment
- Port of Discharge
- The ultimate Destination country and the Purpose of export
- Enter the From Date and To-date under the heading “Period during which item is to be exported.”
- Select the Jurisdictional Regional Authority from the list box
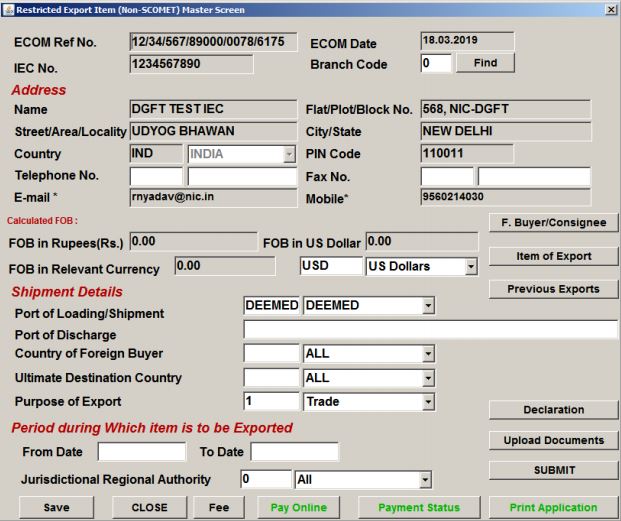 Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Shipment Details
Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Shipment Details
Details of Products to be Exported
After providing the details, click on the Save button to save your details. Select the items of an export button to proceed further. The exporter needs to provide the following details about the products to be exported.
- ITC(HS) code
- Name and Description of Item
- Item Quantity
- Unit of Measurement (UOM)
- FOB in Rupees, FOB in USD, and FOB in relevant Foreign Currency
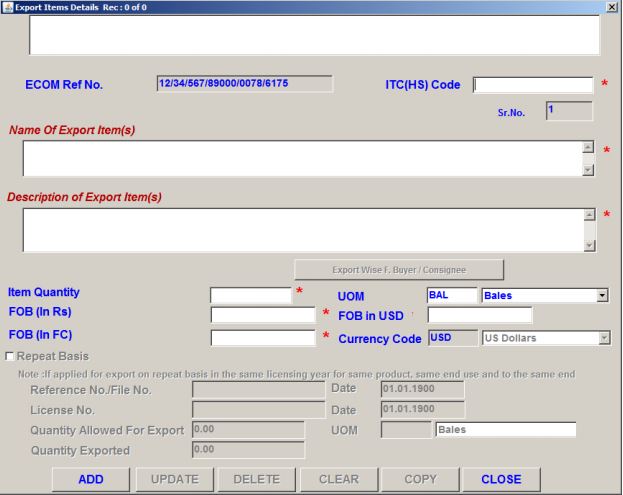 Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Product Details
Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Product Details
Details of foreign Buyer or Consignee
After clicking on the OK button, the exporter needs to fill in the details of the foreign Buyer or consignee. In the next screen, the exporter will have to enter the details of exports of SCOMET Items in the preceding three licensing years.
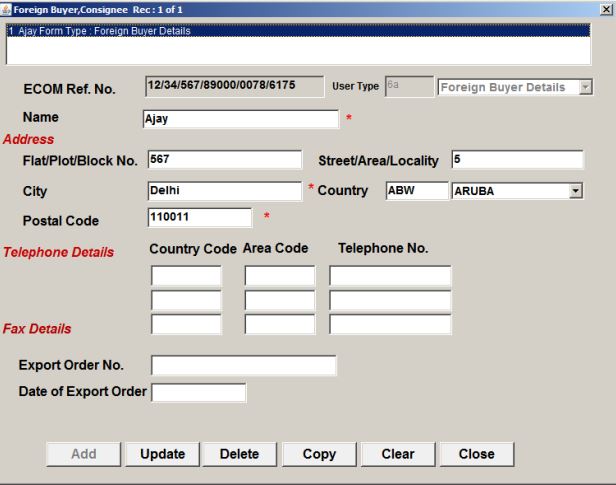 Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Buyer Details
After filling in all the details exporter needs to click on the declaration button on the main screen and furnish the details of Name, Official Address, Telephone, and country details.
Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Buyer Details
After filling in all the details exporter needs to click on the declaration button on the main screen and furnish the details of Name, Official Address, Telephone, and country details.
Upload Document
By clicking on the upload document button, a dialogue box will appear, and the exporter can upload all required documents using the browse button.
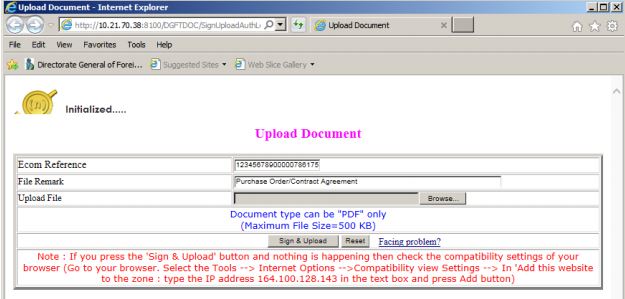 Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Document upload
Once the documents are uploaded successfully, the exporter can apply to DGFT to issue an export license for the shipment of PPE for COVID-19.
Procedure and Criteria for Export of PPE Medical Coveralls for COVID-19 - Document upload
Once the documents are uploaded successfully, the exporter can apply to DGFT to issue an export license for the shipment of PPE for COVID-19.
