Peter
Expert
Published on: Apr 20, 2026
To manufacture or import Active Pharmaceutical Ingredient (API)/drugs to India, the company or the organization should seek prior approval from the Drugs Controller General Of India (DCGI) for licensing to produce and distribute. The company should also follow the norms stipulated by the Central Drugs Standard Control Organisation (CDSCO) to produce the drugs. The Ministry of Health and Family Welfare will monitor the DCGI and CDSCO. To support DCGI and CDSCO in widening scope and research, the Government of India (GoI) has formed the Drug Technical Advisory Board (DTAB) and Drug Consultative Committee (DCC).
Any new S. No. Rule Number Represents 1 2 3 4 The following are also the rules and guidelines that should be followed by the company that introduced new drugs into the market: S. No. Name of the Rules and Guidelines 1 2 3 4 5 6 7 8 9 10 11 To seek permission to manufacture or import new As per rule 21(b), all new The following should be attached while drafting Form 44, applying for approval: The clinical data should include the use of drugs on the following members as per item 8.3 of Appendix I: To Companies importing drugs should register with CDSCO in Form 41. The Form shall contain details of the manufacturing site and drugs used and required. After furnishing in Form-41, the company can approach CDSCO for Import Registration Certificate and Import Licence.
Though registration is not required for clinical tests, New Drug and Clinical Trail Rules, 2019 states that the company imports drugs should furnish details in Form 16 for NOC and Test Licence. For manufacturers in India, the registration fee is Rs.3,00,000
For companies importing drugs, the registration fee is USD 1500
For registering a new drug, the fee is USD 1000 per drug The registration certificate issued by CDSCO is valid for three years from the date issued. To renew the registration certificate, the company should apply with all required documents nine months before the expiry date. To apply for manufacturing, conduct a clinical test, or import new drugs, the applicant should register with CDSCO through an online portal.Manufacturing or Importing API Drugs
Rule 122 A
Permission to Import New Drugs
Rule 122B
Permission to Manufacture New Drugs
Rule 122 DA
Definition of Required Clinical Trials
Rule 122 E
Definition of Drugs
Drugs and Cosmetics Act 1940 and its rules 1945
Narcotic Drugs and Psychotropic Substances of 1985
Drugs Price Control Order of 1995
Consumer Protection Act of 1986
Factories Act of 1948
Law of Contracts (Indian contract Act of 1872)
Monopolistic & Restrictive Trade Practices Act of 1969
ICH GCP Guidelines
Schedule Y Guidelines
ICMR Guidelines
Registry of Trial
Guidelines and Requirements for Approval of Import or Manufacturing New Drugs or Undertaking Clinical Trails
Application and Data Required
Clinical Trails Required for Application Process
Phases of Clinical Trails
Drafting Results for Approval
Studies in Special Populations
Importing New Drugs
Registring with CDSCO
Fees for Registration with CDSCO
List of Forms Required
Validity of the Registration Certificate
Renewal of Registration Certificate
Document Required for Registration
Registering with CDSCO
 Step 2: Click on Login or Sign Up and Click on "Sign Up Here"
Step 2: Click on Login or Sign Up and Click on "Sign Up Here"
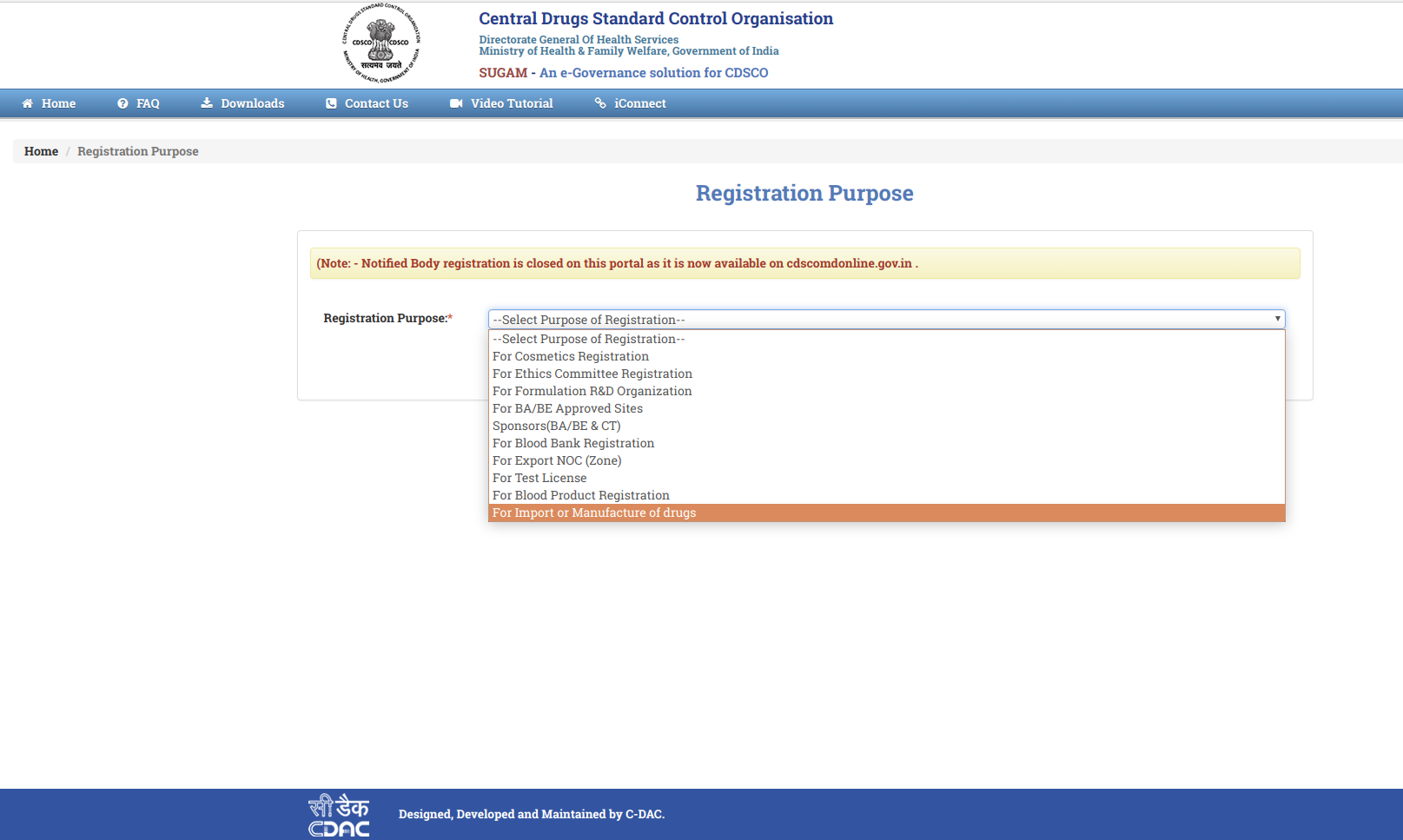
 Step 3: Choose the option from "Registration Purpose" and click, Submit
Step 3: Choose the option from "Registration Purpose" and click, Submit
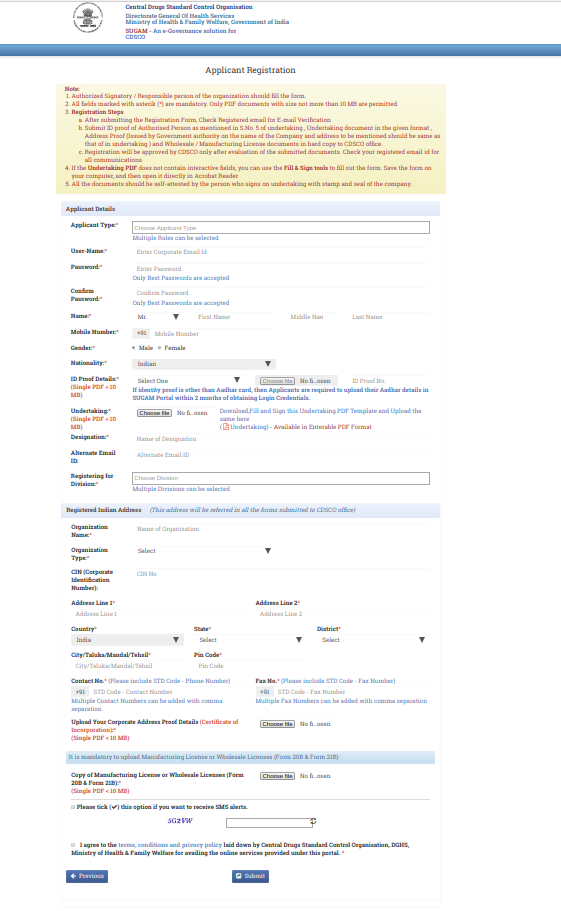
 Step 4: Provide all the details with valid information
Step 4: Provide all the details with valid information
 Step 5: After verification, an e-mail would be sent to the registered e-mail address
Step 6: Create USER ID and Password with the link and login into the CDSCO webpage
Step 7: Click on the Dashboard and click on 'Submit Application' to upload the documents and details for the registration certificate
Step 5: After verification, an e-mail would be sent to the registered e-mail address
Step 6: Create USER ID and Password with the link and login into the CDSCO webpage
Step 7: Click on the Dashboard and click on 'Submit Application' to upload the documents and details for the registration certificate
Applying for Import Licence
Step 1: Login into CDSCO and click on 'Dashboard'
Step 2: Click on 'Application Submission' and click on 'Form Submission' on the popped up window
Step 3: Choose the Department and Select the Form to be submitted
Step 4: (Applicant and Manufacturer Detail): On the 'Type of Application', click 'Fresh'. Fill the details with valid credentials and click on 'Save and Continue'.
Step 5: (Drug Details): Choose the application applying for 'Bulk' or 'Finished Formulation'. Furnish all the details and click on 'Save' and click 'Next'
Step 6: (Manufacturing Unit Details): Enter the details of the manufacturing unit and click on 'Next'. To add more units, click on 'Add'
Step 7: (Directs back to Applicant and Manufacturer Detail): Select the application type as 'Endorsement', furnish all the details, enter the registration number and click on 'Continue'.
Step 8: Verify the form and click on 'Save' and 'Next'
Step 9: Verify the form and click on 'Continue' After clicking 'Continue', the application will be sent for verification and the applicant shall receive File No. for future reference.
Making Payment Online
Step 1: Open CDSCO web portal and log in to CDSCO
Step 2: Click on 'Online Payment Option' and furnish all the required details
Step 3: Choose the Bank
Step 4: Click on Pay, Enter Bank Details and click on 'Proceed'
Step 5: After successful payment, the transaction will generate GAR 6 for e-challan, which can be accessed through NTRP. For more details on MSME, Import and Registration documentation,Eligibility Criteria
